Communication Within and Between Neurons
Thus far, we have described the main characteristics of neurons, including how their processes come in close contact with one another to form synapses. In this section, we consider the conduction of communication within a neuron and how this signal is transmitted to the next neuron. There are two stages of this electrochemical action in neurons. The first stage is the electrical conduction of dendritic input to the initiation of an action potential within a neuron. The second stage is a chemical transmission across the synaptic gap between the presynaptic neuron and the postsynaptic neuron of the synapse. To understand these processes, we first need to consider what occurs within a neuron when it is at a steady state, called resting membrane potential.
Resting Membrane Potential
The intracellular (inside the cell) fluid and extracellular (outside the cell) fluid of neurons is composed of a combination of ions (electrically charged molecules; see Figure 4). Cations are positively charged ions, and anions are negatively charged ions. The composition of intracellular and extracellular fluid is similar to salt water, containing sodium (Na+), potassium (K+), chloride (Cl–), and anions (A–).
The cell membrane, which is composed of a lipid bilayer of fat molecules, separates the cell from the surrounding extracellular fluid. There are proteins that span the membrane, forming ion channels that allow particular ions to pass between the intracellular and extracellular fluid (see Figure 4). These ions are in different concentrations inside the cell relative to outside the cell, and the ions have different electrical charges. Due to this difference in concentration and charge, two forces act to maintain a steady state when the cell is at rest: diffusion and electrostatic pressure. Diffusion is the force on molecules to move from areas of high concentration to areas of low concentration. Electrostatic pressure is the force on two ions with similar charge to repel each other and the force of two ions with opposite charge to attract to one another. Remember the saying, opposites attract?
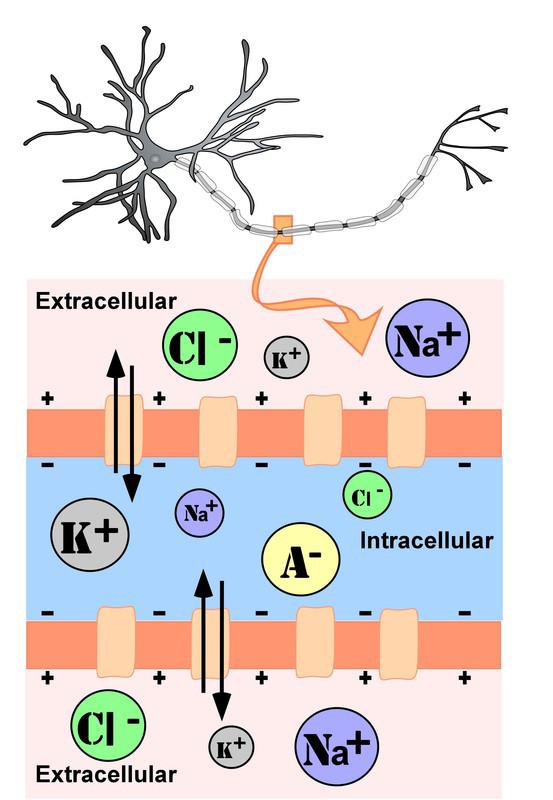
Figure 4. Representation of ion concentrations inside (intracellular) and outside (extracellular) a neuron in the unmylenated segment of the axon.
Regardless of the ion, there exists a membrane potential at which the force of diffusion is equal and opposite of the force of electrostatic pressure. This voltage, called the equilibrium potential, is the voltage at which no ions flow. Since there are several ions that can permeate the cell’s membrane, the baseline electrical charge inside the cell compared with outside the cell, referred to as resting membrane potential, is based on the collective drive of force on several ions. Relative to the extracellular fluid, the membrane potential of a neuron at rest is negatively charged at approximately -70 mV (see Figure 5). These are very small voltages compared with the voltages of batteries and electrical outlets, which we encounter daily, that range from 1.5 to 240 V.
Let us see how these two forces, diffusion and electrostatic pressure, act on the four groups of ions mentioned above.
Anions (A-): Anions are highly concentrated inside the cell and contribute to the negative charge of the resting membrane potential. Diffusion and electrostatic pressure are not forces that determine A– concentration because A– is impermeable to the cell membrane. There are no ion channels that allow for A– to move between the intracellular and extracellular fluid.
Potassium (K+): The cell membrane is very permeable to potassium at rest, but potassium remains in high concentrations inside the cell. Diffusion pushes K+ outside the cell because it is in high concentration inside the cell. However, electrostatic pressure pushes K+ inside the cell because the positive charge of K+ is attracted to the negative charge inside the cell. In combination, these forces oppose one another with respect to K+.
Chloride (Cl-): The cell membrane is also very permeable to chloride at rest, but chloride remains in high concentration outside the cell. Diffusion pushes Cl– inside the cell because it is in high concentration outside the cell. However, electrostatic pressure pushes Cl– outside the cell because the negative charge of Cl– is attracted to the positive charge outside the cell. Similar to K+, these forces oppose one another with respect to Cl–.
Sodium (Na+): The cell membrane is not very permeable to sodium at rest. Diffusion pushes Na+ inside the cell because it is in high concentration outside the cell. Electrostatic pressure also pushes Na+ inside the cell because the positive charge of Na+ is attracted to the negative charge inside the cell. Both of these forces push Na+ inside the cell; however, Na+ cannot permeate the cell membrane and remains in high concentration outside the cell. The small amounts of Na+ inside the cell are removed by a sodium-potassium pump, which uses the neuron’s energy (adenosine triphosphate, ATP) to pump 3 Na+ ions out of the cell in exchange for bringing 2 K+ ions inside the cell.
Action Potential
Now that we have considered what occurs in a neuron at rest, let us consider what changes occur to the resting membrane potential when a neuron receives input, or information, from the presynaptic terminal button of another neuron. Our understanding of the electrical signals or potentials that occurs within a neuron results from the seminal work of Hodgkin and Huxleythat began in the 1930s at a well-known marine biology lab in Woodshole, MA. Their work, for which they won the Nobel Prize in Medicine in 1963, has resulted in the general model of electrochemical transduction that is described here (Hodgkin & Huxley, 1952). Hodgkin and Huxley studied a very large axon in the squid, a common species for that region of the United States. The giant axon of the squid is roughly 100 times larger than that of axons in the mammalian brain, making it much easier to see. Activation of the giant axon is responsible for a withdrawal response the squid uses when trying to escape from a predator, such as large fish, birds, sharks, and even humans. When was the last time you had calamari? The large axon size is no mistake in nature’s design; it allows for very rapid transmission of an electrical signal, enabling a swift escape motion in the squid from its predators.
While studying this species, Hodgkin and Huxley noticed that if they applied an electrical stimulus to the axon, a large, transient electrical current conducted down the axon. This transient electrical current is known as an action potential (see Figure 5). An action potential is an all-or-nothing response that occurs when there is a change in the charge or potential of the cell from its resting membrane potential (-70 mV) in a more positive direction, which is a depolarization (see Figure 5). What is meant by an all-or-nothing response? I find that this concept is best compared to the binary code used in computers, where there are only two possibilities, 0 or 1. There is no halfway or in-between these possible values; for example, 0.5 does not exist in binary code. There are only two possibilities, either the value of 0 or the value of 1. The action potential is the same in this respect. There is no halfway; it occurs, or it does not occur. There is a specific membrane potential that the neuron must reach to initiate an action potential. This membrane potential, called the threshold of excitation, is typically around -50 mV. If the threshold of excitation is reached, then an action potential is triggered.
How is an action potential initiated? At any one time, each neuron is receiving hundreds of inputs from the cells that synapse with it. These inputs can cause several types of fluctuations in the neuron’s membrane potentials (see Figure 5):
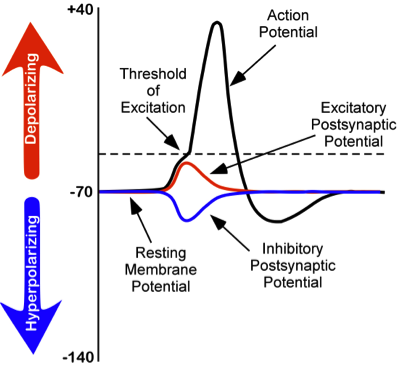
Figure 5. Changes in membrane potentials of neurons.
excitatory postsynaptic potentials (EPSPs): a depolarizing current that causes the membrane potential to become more positive and closer to the threshold of excitation; or inhibitory postsynaptic potentials (IPSPs): a hyperpolarizing current that causes the membrane potential to become more negative and further away from the threshold of excitation.
These postsynaptic potentials, EPSPs and IPSPs, summate or add together in time and space. The IPSPs make the membrane potential more negative, but how much so depends on the strength of the IPSPs. The EPSPs make the membrane potential more positive; again, how much more positive depends on the strength of the EPSPs. If you have two small EPSPs at the same time and the same synapse then the result will be a large EPSP. If you have a small EPSP and a small IPSP at the same time and the same synapse then they will cancel each other out. Unlike the action potential, which is an all-or-nothing response, IPSPs and EPSPs are smaller and graded potentials, varying in strength. The change in voltage during an action potential is approximately 100 mV. In comparison, EPSPs and IPSPs are changes in voltage between 0.1 to 40 mV. They can be different strengths, or gradients, and they are measured by how far the membrane potentials diverge from the resting membrane potential.
I know the concept of summation can be confusing. As a child, I use to play a game in elementary school with a very large parachute where you would try to knock balls out of the center of the parachute. This game illustrates the properties of summation rather well. In this game, a group of children next to one another would work in unison to produce waves in the parachute in order to cause a wave large enough to knock the ball out of the parachute. The children would initiate the waves at the same time and in the same direction. The additive result was a larger wave in the parachute, and the balls would bounce out of the parachute.
However, if the waves they initiated occurred in the opposite direction or with the wrong timing, the waves would cancel each other out, and the balls would remain in the center of the parachute. EPSPs or IPSPs in a neuron work in the same fashion to the properties of the waves in the parachute; they either add or cancel each other out. If you have two EPSPs, then they sum together and become a larger depolarization. Similarly, if two IPSPs come into the cell at the same time, they will sum and become a larger hyperpolarization in membrane potential. However, if two inputs were opposing one another, moving the potential in opposite directions, such as an EPSP and an IPSP, their sum would cancel each other out.
At any moment in time, each cell is receiving mixed messages, both EPSPs and IPSPs. If the summation of EPSPs is strong enough to depolarize the membrane potential to reach the threshold of excitation, then it initiates an action potential. The action potential then travels down the axon, away from the soma, until it reaches the ends of the axon (the terminal button). In the terminal button, the action potential triggers the release of neurotransmitters from the presynaptic terminal button into the synaptic gap. These neurotransmitters, in turn, cause EPSPs and IPSPs in the postsynaptic dendritic spines of the next cell (see Figures 4 & 6). The neurotransmitter released from the presynaptic terminal button binds with ionotropic receptors in a lock-and-key fashion on the post-synaptic dendritic spine. Ionotropic receptors are receptors on ion channels that open, allowing some ions to enter or exit the cell, depending upon the presence of a particular neurotransmitter. The type of neurotransmitter and the permeability of the ion channel it activates will determine if an EPSP or IPSP occurs in the dendrite of the post-synaptic cell. These EPSPs and IPSPs summate in the same fashion described above and the entire process occurs again in another cell.
The Change in Membrane Potential During an Action Potential
We discussed previously which ions are involved in maintaining the resting membrane potential. Not surprisingly, some of these same ions are involved in the action potential. When the cell becomes depolarized (more positively charged) and reaches the threshold of excitation, this causes a voltage-dependent Na+ channel to open. A voltage-dependent ion channel is a channel that opens, allowing some ions to enter or exit the cell, depending upon when the cell reaches a particular membrane potential. When the cell is at resting membrane potential, these voltage-dependent Na+ channels are closed. As we learned earlier, both diffusion and electrostatic pressure are pushing Na+ inside the cells. However, Na+ cannot permeate the membrane when the cell is at rest. Now that these channels are open, Na+ rushes inside the cell, causing the cell to become very positively charged relative to the outside of the cell. This is responsible for the rising or depolarizing phase of the action potential (see Figure 5). The inside of the cell becomes very positively charged, +40mV. At this point, the Na+ channels close and become refractory. This means the Na+ channels cannot reopen again until after the cell returns to the resting membrane potential. Thus, a new action potential cannot occur during the refractory period. The refractory period also ensures the action potential can only move in one direction down the axon, away from the soma. As the cell becomes more depolarized, a second type of voltage-dependent channel opens; this channel is permeable to K+. With the cell very positive relative to the outside of the cell (depolarized) and the high concentration of K+ within the cell, both the force of diffusion and the force of electrostatic pressure drive K+ outside of the cell. The movement of K+ out of the cell causes the cell potential to return back to the resting membrane potential, the falling or hyperpolarizing phase of the action potential (see Figure 5). A short hyperpolarization occurs partially due to the gradual closing of the K+ channels. With the Na+ closed, electrostatic pressure continues to push K+ out of the cell. In addition, the sodium-potassium pump is pushing Na+ out of the cell. The cell returns to the resting membrane potential, and the excess extracellular K+ diffuses away. This exchange of Na+ and K+ ions happens very rapidly, in less than 1 msec. The action potential occurs in a wave-like motion down the axon until it reaches the terminal button. Only the ion channels in very close proximity to the action potential are affected.
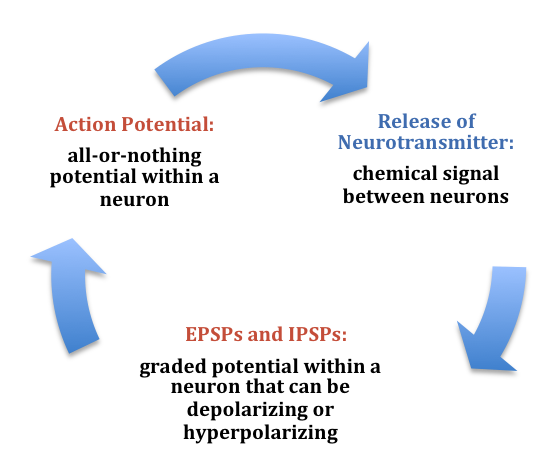
Figure 6. Summary of the electrochemical communication within and between neurons.
Earlier you learned that axons are covered in myelin. Let us consider how myelin speeds up the process of the action potential. There are gaps in the myelin sheaths called nodes of Ranvier. The myelin insulates the axon and does not allow any fluid to exist between the myelin and cell membrane. Under the myelin, when the Na+ and K+ channels open, no ions flow between the intracellular and extracellular fluid. This saves the cell from having to expend the energy necessary to rectify or regain the resting membrane potential. (Remember, the pumps need ATP to run.) Under the myelin, the action potential degrades some, but is still large enough in potential to trigger a new action potential at the next node of Ranvier. Thus, the action potential actively jumps from node to node; this process is known as saltatory conduction.
In the presynaptic terminal button, the action potential triggers the release of neurotransmitters (see Figure 3). Neurotransmitters cross the synaptic gap and open subtypes of receptors in a lock-and-key fashion (see Figure 3). Depending on the type of neurotransmitter, an EPSP or IPSP occurs in the dendrite of the post-synaptic cell. Neurotransmitters that open Na+ or calcium (Ca+) channels cause an EPSP; an example is the NMDA receptors, which are activated by glutamate (the main excitatory neurotransmitter in the brain). In contrast, neurotransmitters that open Cl- or K+ channels cause an IPSP; an example is gamma-aminobutryric acid (GABA) receptors, which are activated by GABA, the main inhibitory neurotransmitter in the brain. Once the EPSPs and IPSPs occur in the postsynaptic site, the process of communication within and between neurons cycles on (see Figure 6). A neurotransmitter that does not bind to receptors is broken down and inactivated by enzymes or glial cells, or it is taken back into the presynaptic terminal button in a process called reuptake, which will be discussed further in the module on psychopharmacology.

