Useful Things to Know about Classical Conditioning
Classical Conditioning Has Many Effects on Behavior
A classical CS (e.g., the bell) does not merely elicit a simple, unitary reflex. Pavlov emphasized salivation because that was the only response he measured. But his bell almost certainly elicited a whole system of responses that functioned to get the organism ready for the upcoming US (food) (see Timberlake, 2001). For example, in addition to salivation, CSs (such as the bell) that signal that food is near also elicit the secretion of gastric acid, pancreatic enzymes, and insulin (which gets blood glucose into cells). All of these responses prepare the body for digestion. Additionally, the CS elicits approach behavior and a state of excitement. And presenting a CS for food can also cause animals whose stomachs are full to eat more food if it is available. In fact, food CSs are so prevalent in modern society, humans are likewise inclined to eat or feel hungry in response to cues associated with food, such as the sound of a bag of potato chips opening, the sight of a well-known logo (e.g., Coca-Cola), or the feel of the couch in front of the television.
Classical conditioning is also involved in other aspects of eating. Flavors associated with certain nutrients (such as sugar or fat) can become preferred without arousing any awareness of the pairing. For example, protein is a US that your body automatically craves more of once you start to consume it (UR): since proteins are highly concentrated in meat, the flavor of meat becomes a CS (or cue, that proteins are on the way), which perpetuates the cycle of craving for yet more meat (this automatic bodily reaction now a CR).
In a similar way, flavors associated with stomach pain or illness become avoided and disliked. For example, a person who gets sick after drinking too much tequila may acquire a profound dislike of the taste and odor of tequila—a phenomenon called taste aversion conditioning. The fact that flavors are often associated with so many consequences of eating is important for animals (including rats and humans) that are frequently exposed to new foods. And it is clinically relevant. For example, drugs used in chemotherapy often make cancer patients sick. As a consequence, patients often acquire aversions to foods eaten just before treatment, or even aversions to such things as the waiting room of the chemotherapy clinic itself (see Bernstein, 1991; Scalera & Bavieri, 2009).
Classical conditioning occurs with a variety of significant events. If an experimenter sounds a tone just before applying a mild shock to a rat’s feet, the tone will elicit fear or anxiety after one or two pairings. Similar fear conditioning plays a role in creating many anxiety disorders in humans, such as phobias and panic disorders, where people associate cues (such as closed spaces, or a shopping mall) with panic or other emotional trauma (see Mineka & Zinbarg, 2006). Here, rather than a physical response (like drooling), the CS triggers an emotion.
Another interesting effect of classical conditioning can occur when we ingest drugs. That is, when a drug is taken, it can be associated with the cues that are present at the same time (e. g., rooms, odors, drug paraphernalia). In this regard, if someone associates a particular smell with the sensation induced by the drug, whenever that person smells the same odor afterward, it may cue responses (physical and/or emotional) related to taking the drug itself. But drug cues have an even more interesting property: They elicit responses that often “compensate” for the upcoming effect of the drug (see Siegel, 1989). For example, morphine itself suppresses pain; however, if someone is used to taking morphine, a cue that signals the “drug is coming soon” can actually make the person more sensitive to pain. Because the person knows a pain suppressant will soon be administered, the body becomes more sensitive, anticipating that “the drug will soon take care of it.” Remarkably, such conditioned compensatory responses in turn decrease the impact of the drug on the body—because the body has become more sensitive to pain.
This conditioned compensatory response has many implications. For instance, a drug user will be most “tolerant” to the drug in the presence of cues that have been associated with it (because such cues elicit compensatory responses). As a result, overdose is usually not due to an increase in dosage, but to taking the drug in a new place without the familiar cues— which would have otherwise allowed the user to tolerate the drug (see Siegel, Hinson, Krank, & McCully, 1982). Conditioned compensatory responses (which include heightened pain sensitivity and decreased body temperature, among others) might also cause discomfort, thus motivating the drug user to continue usage of the drug to reduce them. This is one of several ways classical conditioning might be a factor in drug addiction and dependence.
A final effect of classical cues is that they motivate ongoing operant behavior (see Balleine, 2005). For example, if a rat has learned via operant conditioning that pressing a lever will give it a drug, in the presence of cues that signal the “drug is coming soon” (like the sound of the lever squeaking), the rat will work harder to press the lever than if those cues weren’t present (i.e., there is no squeaking lever sound). Similarly, in the presence of food-associated cues (e. g., smells), a rat (or an overeater) will work harder for food. And finally, even in the presence of negative cues (like something that signals fear), a rat, a human, or any other organism will work harder to avoid those situations that might lead to trauma. Classical CSs thus have many effects that can contribute to significant behavioral phenomena.
[Image courtesy of Bernard W. Balleine]
The Learning Process
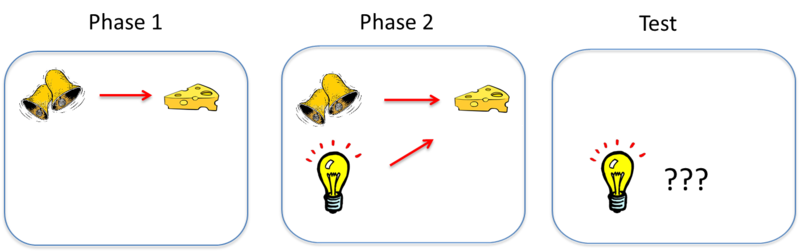
As mentioned earlier, classical conditioning provides a method for studying basic learning processes. Somewhat counterintuitively, though, studies show that pairing a CS and a US together is not sufficient for an association to be learned between them. Consider an effect called blocking (see Kamin, 1969). In this effect, an animal first learns to associate one CS— call it stimulus A—with a US. In the illustration above, the sound of a bell (stimulus A) is paired with the presentation of food. Once this association is learned, in a second phase, a second stimulus—stimulus B—is presented alongside stimulus A, such that the two stimuli are paired with the US together. In the illustration, a light is added and turned on at the same time the bell is rung. However, because the animal has already learned the association between stimulus A (the bell) and the food, the animal doesn’t learn an association between stimulus B (the light) and the food. That is, the conditioned response only occurs during the presentation of stimulus A, because the earlier conditioning of A “blocks” the conditioning of B when B is added to A. The reason? Stimulus A already predicts the US, so the US is not surprising when it occurs with Stimulus B.
Learning depends on such a surprise, or a discrepancy between what occurs on a conditioning trial and what is already predicted by cues that are present on the trial. To learn something through classical conditioning, there must first be some prediction error, or the chance that a conditioned stimulus won’t lead to the expected outcome. With the example of the bell and the light, because the bell always leads to the reward of food, there’s no “prediction error” that the addition of the light helps to correct. However, if the researcher suddenly requires that the bell and the light both occur in order to receive the food, the bell alone will produce a prediction error that the animal has to learn.
Blocking and other related effects indicate that the learning process tends to take in the most valid predictors of significant events and ignore the less useful ones. This is common in the real world. For example, imagine that your supermarket puts big star-shaped stickers on products that are on sale. Quickly, you learn that items with the big star-shaped stickers are cheaper. However, imagine you go into a similar supermarket that not only uses these stickers, but also uses bright orange price tags to denote a discount. Because of blocking (i.e., you already know that the star-shaped stickers indicate a discount), you don’t have to learn the color system, too. The star-shaped stickers tell you everything you need to know (i.e. there’s no prediction error for the discount), and thus the color system is irrelevant.
Classical conditioning is strongest if the CS and US are intense or salient. It is also best if the CS and US are relatively new and the organism hasn’t been frequently exposed to them before. And it is especially strong if the organism’s biology has prepared it to associate a particular CS and US. For example, rats and humans are naturally inclined to associate an illness with a flavor, rather than with a light or tone. Because foods are most commonly experienced by taste, if there is a particular food that makes us ill, associating the flavor (rather than the appearance—which may be similar to other foods) with the illness will more greatly ensure we avoid that food in the future, and thus avoid getting sick. This sorting tendency, which is set up by evolution, is called preparedness.
There are many factors that affect the strength of classical conditioning, and these have been the subject of much research and theory (see Rescorla & Wagner, 1972; Pearce & Bouton, 2001). Behavioral neuroscientists have also used classical conditioning to investigate many of the basic brain processes that are involved in learning (see Fanselow & Poulos, 2005; Thompson & Steinmetz, 2009).
Erasing Classical Learning
After conditioning, the response to the CS can be eliminated if the CS is presented repeatedly without the US. This effect is called extinction, and the response is said to become “extinguished.” For example, if Pavlov kept ringing the bell but never gave the dog any food afterward, eventually the dog’s CR (drooling) would no longer happen when it heard the CS (the bell), because the bell would no longer be a predictor of food. Extinction is important for many reasons. For one thing, it is the basis for many therapies that clinical psychologists use to eliminate maladaptive and unwanted behaviors. Take the example of a person who has a debilitating fear of spiders: one approach might include systematic exposure to spiders. Whereas, initially the person has a CR (e.g., extreme fear) every time s/he sees the CS (e.g., the spider), after repeatedly being shown pictures of spiders in neutral conditions, pretty soon the CS no longer predicts the CR (i.e., the person doesn’t have the fear reaction when seeing spiders, having learned that spiders no longer serve as a “cue” for that fear). Here, repeated exposure to spiders without an aversive consequence causes extinction.
Psychologists must accept one important fact about extinction, however: it does not necessarily destroy the original learning (see Bouton, 2004). For example, imagine you strongly associate the smell of chalkboards with the agony of middle school detention. Now imagine that, after years of encountering chalkboards, the smell of them no longer recalls the agony of detention (an example of extinction). However, one day, after entering a new building for the first time, you suddenly catch a whiff of a chalkboard and WHAM!, the agony of detention returns. This is called spontaneous recovery: following a lapse in exposure to the CS after extinction has occurred, sometimes re-exposure to the CS (e.g., the smell of chalkboards) can evoke the CR again (e.g., the agony of detention).
Another related phenomenon is the renewal effect: After extinction, if the CS is tested in a new context, such as a different room or location, the CR can also return. In the chalkboard example, the action of entering a new building—where you don’t expect to smell chalkboards —suddenly renews the sensations associated with detention. These effects have been interpreted to suggest that extinction inhibits rather than erases the learned behavior, and this inhibition is mainly expressed in the context in which it is learned (see “context” in theKey Vocabulary section below).
This does not mean that extinction is a bad treatment for behavior disorders. Instead, clinicians can increase its effectiveness by using basic research on learning to help defeat these relapse effects (see Craske et al., 2008). For example, conducting extinction therapies in contexts where patients might be most vulnerable to relapsing (e.g., at work), might be a good strategy for enhancing the therapy’s success.

